Why Diamonds and Graphite Are the Same Element
 Carbon can be soft or hardest, depending on structure.
Carbon can be soft or hardest, depending on structure.
Same atoms, different worlds
By Peter Teoh, Science Writer
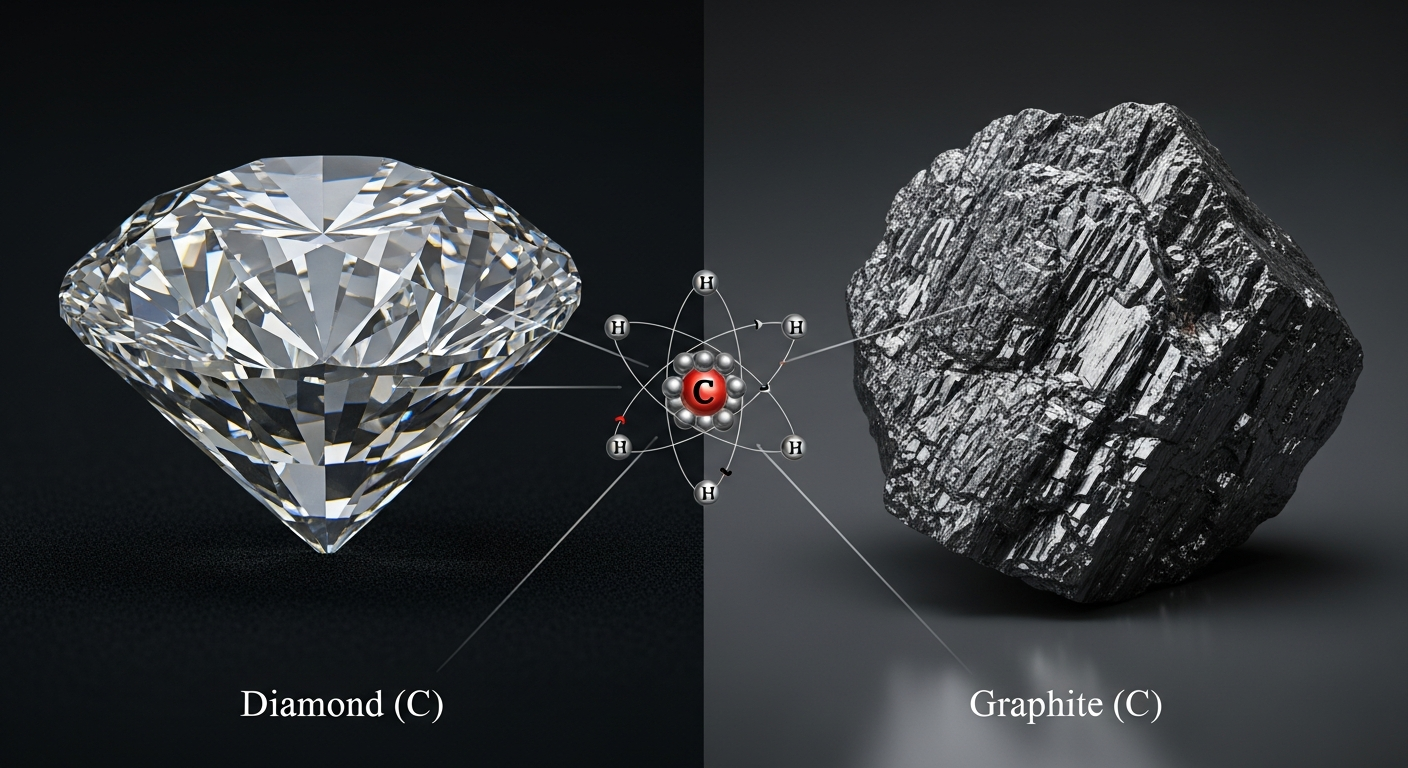
Diamonds and graphite are both pure carbon. Their very different properties come from how atoms are arranged and bonded.
Explainer: Structure drives properties
Focus: In diamond, each carbon atom bonds to four neighbors in a rigid 3D lattice. This makes diamond extremely hard and transparent.
In graphite, carbon atoms form flat sheets with weak forces between layers. The layers slide easily, which is why graphite is soft and conducts electricity.
Summary of Key Ideas:
- Allotropes are different structures of the same element.
- Diamond is a 3D lattice; graphite is layered sheets.
- Bonding explains hardness and conductivity.
Side Notes
- Graphene is a single layer of graphite.
- Carbon also forms buckyballs and nanotubes.
Trending Sidebar
- Graphene electronics and materials.
- Synthetic diamond manufacturing.
Leave a comment